Research
Structural cell biology of bacterial biofilm formation
Rather than living as single, isolated cells in monocultures most bacteria on this planet form macroscopic, surface-attached, multi-cellular communities known as biofilms. Bacterial biofilms play a role in many diseases and infectious processes such as cystic fibrosis and urinary tract infections. Understanding this fundamental bacterial developmental state is therefore critical to understand the colonization process of pathogenic bacteria. The goal of the lab is to use structural biology techniques, coupled with high-resolution imaging to study general principles governing bacterial biofilm formation. We use electron cryomicroscopy (cryo-EM) and tomography (cryo-ET) to resolve structures of molecules that mediate biofilm formation. Correlated light and electron microscopy (CLEM) and mass spectrometry (MS) are used to support our investigations. We combine in vitro reconstitution of key molecules with in vivo imaging to understand how pathogenic bacteria including (but not limited to) Pseudomonas aeruginosa and Escherichia coli form biofilms. Please click on the cartoon for more details (Image Credit: Mitch Blunt for the Wellcome Trust).
Developing structural imaging and image processing techniques
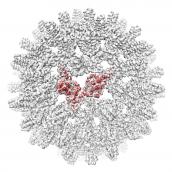
One of the central tools for investigating biological material (cells, tissues and whole organisms) is microscopy. Fine details of the specimen under investigation may be visualized at high resolution using modern microscopes. It is our goal to develop and apply the latest electron microscopy imaging technology to study the important biomedical problem of bacterial biofilms. Most specifically, we are interested in developing methods for in situ macromolecular structure determination using cryo-ET. We are equipped with the latest generation electron microscopes (Titan Krios / Talos Arctica) fitted with advanced direct electron detectors (Falcon 4, K3) in the Central Oxford Structural and Molecular Imaging Centre. Instruments are optimised for high-throughput single-particle as well as tomographic data collection. We have various pieces of supporting equipment, including cryo-light microscopes, cryo-FIB-SEM, a dedicated computing cluster for image processing, as well as a room temperature EM facility with high-pressure freezers and ultramicrotomes. Please click the icosahedral cryo-EM density for more details.
Prokaryotic S-layers
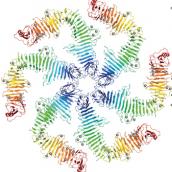
We leverage our structural biology method development to study how prokaryotic cells are encapsulated by surface layers (S-layers). S-layers play critical roles in protecting cells from predators, maintaining cell shape and in promoting biofilm formation. In particular, we use in situ cryo-ET, in conjuction with single-particle cryo-EM, X-ray crystallography and cryo-CLEM to study the structural dynamics of S-layers during the prokaryotic cell cycle. Please click the Caulobacter crescentus SLP X-ray structure for more details.
Funding
Our work is supported by the Wellcome Trust (https://wellcome.ac.uk), the Royal Society (https://royalsociety.org/), the Vallee foundation (https://www.thevalleefoundation.org/), Action Medical Research (https://www.action.org.uk/), Cystic Fibrosis Trust, (https://www.cysticfibrosis.org.uk/), European Molecular Biology Organization (http://www.embo.org), the Leverhulme Trust, BBSRC, MRC, the EPA Research Fund, EP Cephalosporin Fund, the Sir William Dunn School of Pathology and the University of Oxford.